Scientists have uncovered new insights into the biological mechanisms of healthy ageing, showing that the cells surrounding stem cells play a decisive role in how tissues age and lose their regenerative capacity. The findings shift the focus of ageing research from stem cells alone to the health of their supporting cellular environment, opening promising new avenues for interventions aimed at preserving tissue function over time.
The study was conducted by researchers at the Agharkar Research Institute, an autonomous institute under the Department of Science and Technology. Using the fruit fly Drosophila melanogaster as a model system, the team investigated how reproductive stem cells are maintained throughout life and why their function declines with age. The research has been published as a cover article in the journal Stem Cell Reports.
Healthy ageing is a global scientific priority, as populations worldwide seek ways to delay age related decline in tissues such as skin, muscle, intestine and reproductive organs. While previous research has largely focused on damage accumulating within stem cells themselves, the new study demonstrates that ageing may instead begin in the stem cell microenvironment, also known as the stem cell niche.
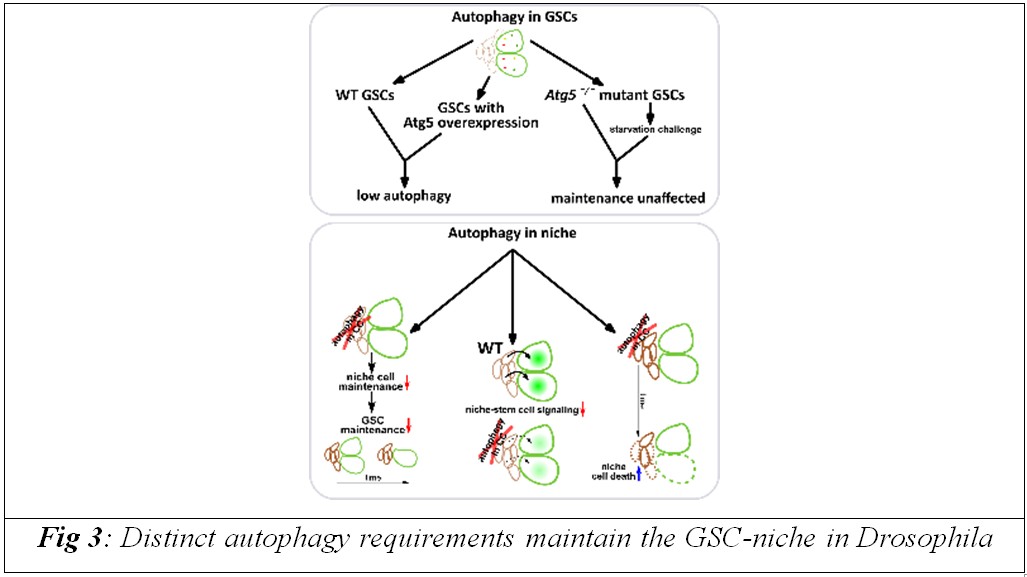
The researchers found that germline stem cells, which are responsible for continuously producing eggs in the ovary, are surprisingly resilient and can function with very low levels of autophagy, the cell’s internal recycling and repair system. In contrast, neighbouring support cells known as cap cells were found to be highly dependent on autophagy for their long term survival and structural integrity.
When key autophagy related genes, including Atg1, Atg5 and Atg9, were selectively disabled in cap cells, these niche cells began to accumulate damage, lost their organised structure and gradually failed to provide essential maintenance signals to the stem cells. Although the stem cells themselves initially remained intact, they were eventually lost because their supportive microenvironment collapsed.
Cap cells play a crucial role in maintaining stem cell identity by continuously supplying biochemical cues, including Bone Morphogenetic Protein signals. The study shows that as autophagy declines in these niche cells during midlife, BMP signalling weakens, directly linking deterioration of the microenvironment to the loss of tissue regenerative capacity.
The findings challenge the long held view that ageing is driven primarily by intrinsic damage within individual stem cells. Instead, the study highlights ageing as a community level process, where the fate of stem cells is closely tied to the health of surrounding support cells. Different cell types within the same tissue were shown to have distinct requirements for autophagy, underscoring the importance of studying entire cellular ecosystems rather than isolated cell populations.
Led by Kiran Suhas Nilangekar and Bhupendra V Shravage of the Developmental Biology Group at ARI Pune, the research places the institute at the forefront of studies on stem cell niche ageing. The work identifies support cells as early weak links in ageing tissues, capable of triggering functional decline even when stem cells remain relatively robust.
Although conducted in fruit flies, the biological pathways examined in the study, including autophagy and stem cell niche signalling, are highly conserved across species. As a result, the findings are expected to inform future research on mammalian tissues such as the intestine, skin and muscle, where similar relationships between stem cells and their niches exist.
The researchers suggest that strategies aimed at strengthening or protecting support cells could indirectly preserve stem cell function, offering new directions for interventions to maintain fertility and tissue health during ageing. Future studies will explore how different cell types balance resilience and vulnerability over time, and whether targeted modulation of autophagy in niche cells can slow age related loss of regenerative capacity.